|
5/5/2023 0 Comments Halogen reactivityTwo experiments with and without ACEIC in the model were established, and their results were compared with each other. In this study, we conducted numerical simulations for January, April, July and October 2015 by using the Community Multiscale Air Quality (CMAQ) modeling system with updated heterogeneous reactions of nitrogen oxides with particulate chlorine and updated Anthropogenic Chlorine Emission Inventory for China (ACEIC). However, its seasonal impacts on surface ozone levels in China have yet been comprehensively explored. Previous models showed negative dependences that we attribute to erroneous titration of OH by isoprene.Īnthropogenic chlorine emission is an important source of Cl radicals, which plays an important role in the oxidative chemistry of the troposphere. We find that the dependence of surface ozone on isoprene emission is positive throughout the U.S., even if NOx emissions are reduced by a factor of 4. ∑ANs can provide an important reservoir for exporting NOx from the U.S. 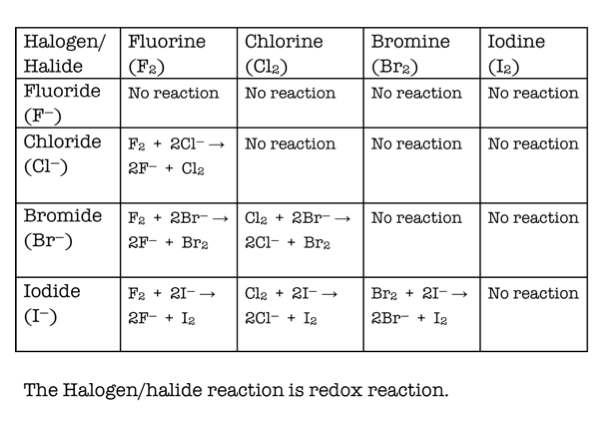 
The correlations of ∑ANs with HCHO and ozone then provide sensitive tests of isoprene chemistry and argue in particular against a fast isomerization channel for isoprene peroxy radicals. ∑ANs in the model is principally composed of secondary isoprene nitrates, including a major contribution from nighttime isoprene oxidation. The model successfully reproduces the observed concentrations of organic nitrates (∑ANs) and their correlations with HCHO and ozone. Simulation of the ozone-CO correlation is improved relative to previous versions of the model, and this is attributed to a lower and reversible yield of isoprene nitrates, increasing the ozone production efficiency per unit of nitrogen oxides (NOx ≡ NO + NO2). The model achieves an unbiased simulation of ozone in the boundary layer and the free troposphere, reflecting canceling effects from recent model updates for isoprene chemistry, bromine chemistry, and HO2 loss to aerosols. Model results are evaluated with observations for ozone, isoprene oxidation products, and related species from the International Consortium for Atmospheric Research on Transport and Transformation aircraft campaign over the eastern United States in summer 2004. We implement a new isoprene oxidation mechanism in a global 3-D chemical transport model (GEOS-Chem). In the case of HI we detected the primary product I as well as the secondary product IO formed in the reaction of NO3 with I. The secondary products Br2 and I2, owing to the heterogeneous recombination of halogen atoms, were observed. In the presence of a concurrent flow of HBr and HI the uptake coefficients of NO3 on ice were found to be high with γ=0.35☐.08 for HBr and γ=0.55☐.1 for HI. In the presence of HCl an upper limit for the uptake of NO3 on ice of γ<1×10-4 was determined and no products were observed. The uptake of NO3 on ice in the presence of HX was studied at 190 K. The reaction of NO3 with NaCl resulted in HCl which was the sole observed product. We observed Br2 and I2 as secondary products resulting from heterogeneous recombination of Br and I, respectively. 
For KBr and KI the primary products Br and I were detected. 
The solid salt substrate contains approximately 10 formal monolayers of adsorbed H2O which enables the ionic exchange reaction. The uptake of NO3 on solid salt at ambient temperature was found to be fast with an uptake coefficient of γ=(4.6±4)×10-2 for NaCl, γ=0.2☐.1 for KBr and γ=0.33☐.09 for KI and NaI. The heterogeneous reactions of NO3 on salt (NaCl, KBr, KI and NaI) and on ice in the presence of HX (X=Cl, Br and I) were studied using a Knudsen cell reactor.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
